38 5.1 models of the atom worksheet answers
Prentice Hall Chemistry Chapter 5.1 Flashcards | Quizlet Prentice Hall Chemistry Chapter 5.1. Term. 1 / 4. Atomic Orbital. Click the card to flip 👆. Definition. 1 / 4. A mathematical expression describing the probability of finding an electron at various locations; usually represented by the region of space around the nucleus where there is a high probability of finding an electron. Click the card ... Unit 3: Atomic Structure & the Periodic Table - Google Bohr Model Bohr Model Worksheet Part 3: Atomic Structure & Theory & Periodic Table PPT Guided Notes Atomic Structure Worksheets Test 3 (Chapter 4 &5): Thursday Feb. 23rd Friday 2/18/2017 19: Chapter 5: Periodic Table: Bell Ringer The Atoms Family Atoms & the Periodic Table: Test 3 (Chapter 4 &5): Friday Feb. 24th Tuesday 2/21/2017 20
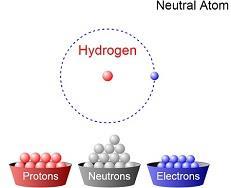
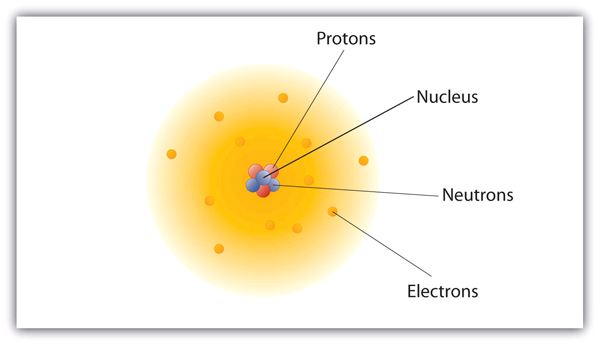
PDF NATURAL SCIENCES - GRADE 8 Term 2: Matter and Materials Unit 1: Atoms ... Worksheet - Atoms 1. Give the following definitions: 1.1 Atoms: 1.2 Matter: 2. Give one word for the following: 2.1 Particles with a negative charge. 2.2 Particles without a charge that are found in the nucleus. 2.3 Particles that determine the volume of an atom. 2.4 Particles responsible for the mass of an atom.

5.1 models of the atom worksheet answers
PDF Chemistry: Matter and Change - Mr. McKnight Clawson High School 5.1 Light and Quantized Energy The Wave Nature of Light, (cont.) , • Sunlight contains a continuous range of wavelengths and frequencies. • A prism separates sunlight into a continuous spectrum of colors. • The electromagnetic spectrumincludes all forms of electromagnetic radiation. SECTION, 5.1 Light and Quantized Energy The Wave Nature of Light, PDF 1.#Read#179*186 2.#Page#186##1,#2,#5,#6,#8 3.#Answer#questions#on#worksheet LearningGoals • Icandescribe(the(components(of(atoms(and theirlocationswithinatoms. • I(can(describe(how(the(work(of(Dalton, Thomson,Rutherford(and(Bohr ... PDF Example Exercise 5.1 Atomic Notation - austincc.edu The maximum number of electrons in the third energy level is found by adding the three sublevels together: 3s+ 3p+ 3d= total electrons, 2 e-+ 6 e-+ 10 e-= 18 e-, The third energy level can hold a maximum of 18 electrons. Of course, in elements where the third energy level of an atom is not filled, there are fewer than 18 electrons.
5.1 models of the atom worksheet answers. PDF Weebly The lowest allowable energy state of an atom is called its 2. Bohr's model of the atom predicted the hydrogen's atomic emission spectrum. frequencies 3. According to Bohr's atomic model, the smaller an electron's orbit, the lower the atom's energy level. 4. PPTX PowerPoint Presentation If you feel that your students can explain methane burning and answer the Three Questions, skip to activity 5.2. Divide the class into pairs and give each pair a molecular model kit, a set of Forms of Energy Cards, and Molecular Models 11 x 17 Placemat. Pass out one copy of 5.1 Molecular Models for Methane Worksheet to each student. 1.1 Simple Model of the Atom - Save My Exams 1.1 Simple Model of the Atom, Medium, Download PDF Quick Answers, 1 2 3 4 5, Question 1, Marks: 1, The atomic structure of an unknown element is shown below. What is the atomic number and mass number for the unknown element? Choose your answer, A, B, C, D, View Answer, Next Question, Unit Resources - Ms. Hart's Chemistry Class 2013-2014 3.3 - History of the Atom I Thomson - Powerpoint - Reading - Guided Notes 3.4 ... 4.2 Bohr Model and Electron Configuration - Powerpoint - Guided Notes 4.3 Valence Electrons ... Powerpoint - Worksheet - ANSWERS!! Page 1 - Page 2 NAMING CHEAT SHEET for EXTRA HELP!
Answer Key Chapter 3 - Chemistry: Atoms First 2e | OpenStax The Bohr model assumes that the electrons move in circular orbits that have quantized energies, angular momentum, and radii that are specified by a single quantum number, n = 1, 2, 3, …, but this quantization is an ad hoc assumption made by Bohr to incorporate quantization into an essentially classical mechanics description of the atom. Bohr ... PDF Answer Key Worksheet 5.1 Naming and Writing Ionic Compounds with ... 17. cesium hydrogen sulfite, 18. mercury (II) phosphite, 19. iron (III) oxalate, 20. barium perchlorate, 21. lithium phosphite, 22. ammonium oxalate, Fe(C2H3O2)2, Al2(Cr2O7)3, Ag2SO4, Sn(NO2)4, Sr(ClO3)2, CsHSO3, Hg3(PO3)2, Fe2(C2O4)3, Ba(ClO4)2, Li3(PO3) (NH4)2C2O4, 23. tin (IV) sulfate, 24. silver dichromate, 25. lead (IV) permanganate, Acces PDF Chapter 5 Electrons In Atoms Assessment Answers Chapter 5 - Electrons in Atoms Section 5.1 - Models of the Atom The Rutherford's model of the atom, did not explain how an atom can emit light or the chemical properties of an atom. This video describes light as a particle and wave. It also describes matter and quantum of energy. PDF Livingston Public Schools / LPS Homepage Use each of the terms below to complete the statements. 1. The lowest allowable energy state of an atom is called its 2. Bohr's model of the atom predicted the hydrogen's atomic emission spectrum. 3. According to Bohr's atomic model, the smaller an electron's orbit, the the atom's energy level. 4.
Get Free Chapter 5 Electrons In Atoms Assessment Answers sublevels in an energy level is equal to the square of the principal, quantum number of that energy level. 12. The maximum number, of electrons that can occupy the fourth principal energy level of, an atom is 32. 13. The higher the energy level occupied by an, electron the more, cardinalnewman.enschool.org, PDF Chapter 5.1 Revising the Atomic Model - Mr.Nguyen's Pre AP Chemistry Chapter 5.1 Revising the Atomic Model, Page 140 5.1 Lesson Check #1-7, 1. The Bohr model propose that an electron is found only in specific circular paths or orbits around the nucleus. Electrons in Bohr's model have specific energies. These specific energies of an electron are called energy levels. 2. Worksheet 5.1 - Models of the Atom.pdf - Course Hero View Homework Help - Worksheet 5.1 - Models of the Atom.pdf from ECON 101 at Omni College. Models of the Atom WS 5.1 Name:_ Per:_ Part I - Complete the following table of atomic models Name of Solved 5.1 Stereochemistry Worksheet Naming Chirality | Chegg.com 5.1 Stereochemistry Worksheet Naming Chirality Centers - Rands Hipso 14 is back CHE = 0 Hi CH,CHE CH.CH . . Which group in the model above has the highest priority? (Circle one) / CH / CH CH / Br Which group in the model above has the lowest priority? (Circle ane) H/CH/CHCH,/Br Which atom from the groups in the model has the highest atomic number?
PDF CHAPTER 6: Earth science Answers Atom and mass conservation 1. ... The following diagram models a chemical reaction but the equation is not balanced. (a) Explain why this equation is not balanced. ... Worksheet 5.1 Science Quest 10: pages 199-201 Carbon monoxide; water Reactants: C = 1; O = 2; H = 2 Products: C = 1; O = 2; H = 2 Reactant mass = (12 + 16 + 16) + (1 ...
Science Chem/Physics - Mr. O'Leary / Regents Physics HW #1: Finish Duality and Models of the atom worksheet. HW #2: Finish Mass and Energy relationship worksheet. Labs and Related Links. Modern Physics Website Activity . Bill Nye Theory of relativity video.14min . Bill Nye Subatomic Physics video.14 min .
The Mechanical Universe of Phyz The Atom [TpT Questions] 50. Particles and Waves [TpT ... Models of the Atom Video 7.3. Wave-Particle Duality: QUESTION SET 8: FORCES AND RELATIVITY Video 8.1. The Fundamental Forces Video 8.2. The Michelson-Morley Experiment ...
CK-12 Chemistry - Basic Answer Key Chapter 5: Electrons in Atoms Answers , 1. The antenna shown above on the left is designed to receive shortwave radio , waves, while the antenna shown above on the left is designed to receive , long-wave radio waves - like that use for CB-radio transmission. 2. Black lights refer to lights which emit UV radiation, which is invisible. They ,
Balbharati solutions for General Science 8th Standard Maharashtra State ... Balbharati solutions for General Science 8th Standard Maharashtra State Board chapter 5 (Inside the Atom) include all questions with solution and detail explanation. This will clear students doubts about any question and improve application skills while preparing for board exams. The detailed, step-by-step solutions will help you understand the concepts better and clear your confusions, if any.
5.1 The Nuclear Model of the Atom - Save My Exams Question 1, Marks: 1, Choose your answer, A, B, C, D, View Answer, Next Question, 1. Motion, Forces & Energy, 2. Thermal Physics, 3. Waves, 4. Electricity & Magnetism, 5. Nuclear Physics, 5.1 The Nuclear Model of the Atom, Multiple Choice Questions, 5.2 Radioactivity, 6. Space Physics,
PDF I. Model Problems. II. Practice Problems III. Challenge questions IV ... I. Model Problems , Multiplying numbers written in Scientific Notation is a three step process. 1. Group the coefficients and exponents together , (1.5 x 103) x (7.3 x 102) = (1.5 x 7.3) x (103x 102) , 2. Multiply the coefficients and add the exponents , (1.5 x 7.3) x (103x 102) = (10.95 x 105) , 3. Convert result to scientific notation ,
5.1 Models of the Atom Section Review Flashcards | Quizlet Start studying 5.1 Models of the Atom Section Review. Learn vocabulary, terms, and more with flashcards, games, and other study tools.
Download Free Chapter 5 Electrons In Atoms Assessment Answers Chapter 5: Electrons in Atoms Models of the Atom Rutherford used existing ideas about the atom, and proposed an atomic model in which the electrons move around the nucleus, like the planets, move around the sun. Section 5.2 - Electron Arrangement in Atoms The electron configuration of an atom is the arrange-, ment of the electrons.
PDF 5 ATOMIC STRUCTURE - National Institute of Open Schooling On this basis he proposed a model for the structure of atom. According to his model, atoms can be considered as a large sphere of uniform positive charge with a number of small negatively charged electrons scattered throughout it, Fig. 5.4. This model was called as plum pudding model. The electrons represent the plums in the pudding made of ...
Chemistry (12th Edition) Chapter 5 - Electrons in Atoms - GradeSaver Chapter 5 - Electrons in Atoms - 5.1 Revising the Atomic Model - 5.1 Lesson Check - Page 132: 5, Answer, Quantized energies means that electrons are moved between energy levels by gaining or losing a certain amount of energy. Work Step by Step,
PDF Example Exercise 5.1 Atomic Notation - austincc.edu The maximum number of electrons in the third energy level is found by adding the three sublevels together: 3s+ 3p+ 3d= total electrons, 2 e-+ 6 e-+ 10 e-= 18 e-, The third energy level can hold a maximum of 18 electrons. Of course, in elements where the third energy level of an atom is not filled, there are fewer than 18 electrons.
PDF 1.#Read#179*186 2.#Page#186##1,#2,#5,#6,#8 3.#Answer#questions#on#worksheet LearningGoals • Icandescribe(the(components(of(atoms(and theirlocationswithinatoms. • I(can(describe(how(the(work(of(Dalton, Thomson,Rutherford(and(Bohr ...
PDF Chemistry: Matter and Change - Mr. McKnight Clawson High School 5.1 Light and Quantized Energy The Wave Nature of Light, (cont.) , • Sunlight contains a continuous range of wavelengths and frequencies. • A prism separates sunlight into a continuous spectrum of colors. • The electromagnetic spectrumincludes all forms of electromagnetic radiation. SECTION, 5.1 Light and Quantized Energy The Wave Nature of Light,






























0 Response to "38 5.1 models of the atom worksheet answers"
Post a Comment