44 charting oxidation number worksheet answers
chapter 7 section 2 oxidation numbers Flashcards | Quizlet what is oxygens oxidation number? -2 unless it is combines with F which means it will be +2 or if it is in a peroxide (H2O2 or Na2O2) when it is -1, what is the oxidation number of hydrogen, +1 unless it is combined with a metal in which case it is -1, what are the oxidation numbers of group 1, 2, and aluminum, +1, +2, and +3, Oxidation Number - Definition, Calculation, Examples, Problems ... - BYJUS Example 2: Oxidation number of Manganese in permanganate ion MnO4-. Charge on the permanganate ion is -1. Oxidation state of permanganate ion =Oxidation state of manganese + 4 oxidation state of oxygen = -1. Oxidation state of oxygen = -2. Oxidation states → x + (4*-2) = -1: x = +7. Atoms in the species → Mn 4O.
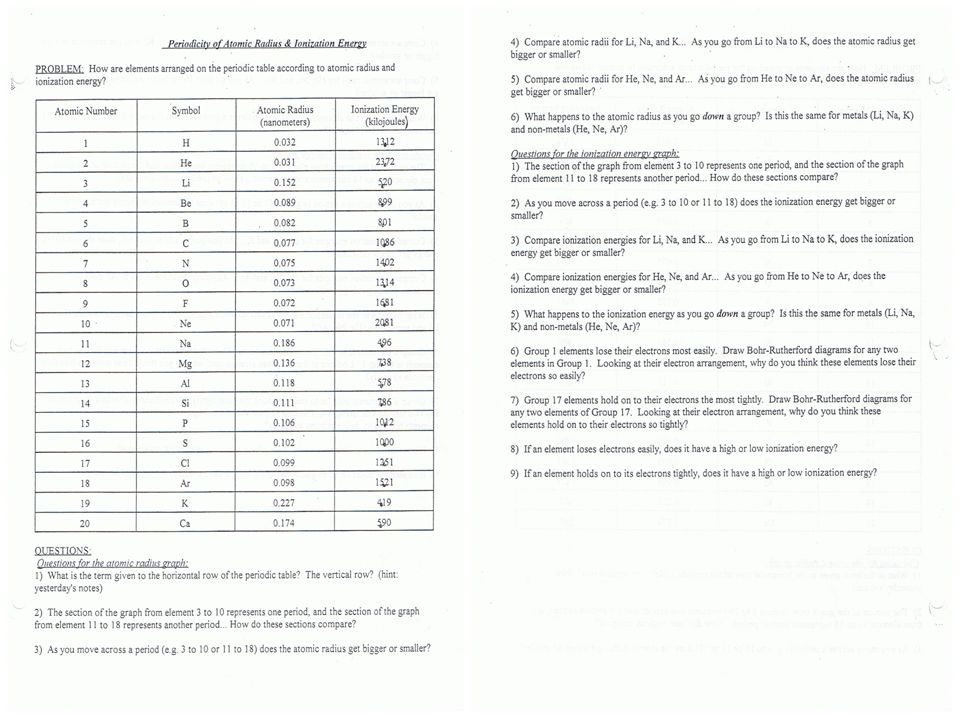
PDF WORKSHEET 1: Determination of oxidation number or valence number Using your periodic table, which shows the common oxidation states of the e lements, and tables in your textbook that show the oxidation state of polyatomic ions, check the total oxidation states of the compounds on this page. If the total oxidation state equals zero, write "CORRECT" in the blank.
Charting oxidation number worksheet answers
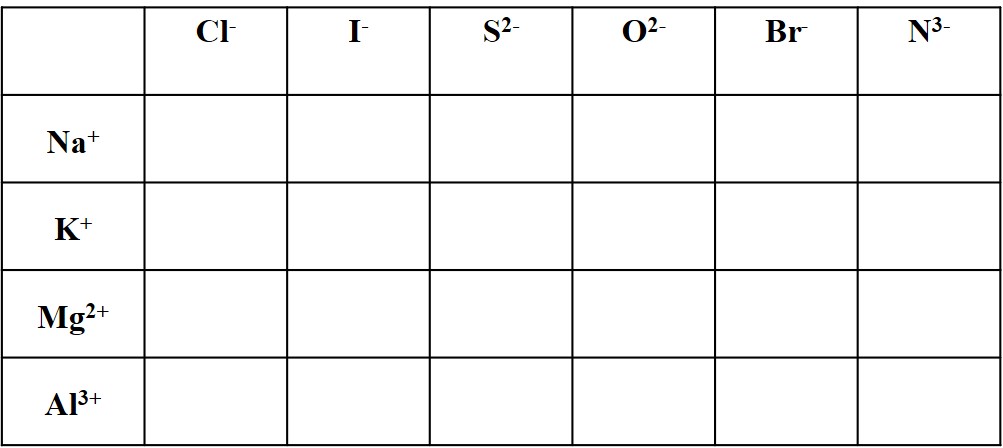
Assigning Oxidation Numbers - Key - WORKSHEET - StuDocu Oxidation answers! worksheet assigning oxidation numbers name period oxidation number rules: pure element has an oxidation number of the oxidation number of an ... STI Chart SP2019; Books. Chemistry: The Central Science; ... Oxidation answers! University Syracuse University; Course Earth Science (EAR 105 ) Academic year 2016/2017; PDF RULES FOR ASSIGNING OXIDATION NUMBERS - California State University ... The oxygen appears to have "lost" 2 electrons, so its oxidation number is +2.* *When compared to the electrically neutral atom. 4. (a) The metallic element in an ionic compound has a positive oxidation number. For monoatomic cations, the oxidation number is equal to the charge on the ion. For example, Na+, Ca2+, Al 3+, Fe , etc. PDF Oxidation Numbers: Rules - Texas A&M University Oxidation Numbers: Rules 1) The oxidation number of the atoms in any free, uncombined element, is zero 2) The sum of the oxidation numbers of all atoms in a compound is zero 3) The sum of the oxidation numbers of all atoms in an ion is equal to the charge of the ion 4) The oxidation number of fluorine in all its compounds is -1
Charting oxidation number worksheet answers. Charting Oxidation Number Worksheet Answers - Fill Online, Printable ... Get the free charting oxidation number worksheet answers form, Description of charting oxidation number worksheet answers, Bonding Basics Ionic Bonds Name Complete the chart for each element. Element # of Protons # of Electrons # of Valence Electrons Sodium Chlorine Beryllium Fluorine Lithium Oxygen Phosphorus Follow, charting oxidation number worksheet answers 33 Charting Oxidation Number Worksheet Answers - Worksheet Project List isme-special.blogspot.com. oxidation charting. Piano Keyboard Worksheet Teachers Pay Theory - Waterandwine.co ... 31 Oxidation Numbers Worksheet Answers - Worksheet Information nuviab6ae4.blogspot.com. oxidation. Printable Bar Graph Worksheets - Worksheet For Kindergarten oxidation state worksheet answers 32 Oxidation Reduction Worksheet Answers - Support Worksheet martindxmguide.blogspot.com. oxidation reduction clo2 aq cuso. 10 Best Images Of Air Pollution Worksheet - Cartoon Refrigerator Clip . oxidation charting answers number worksheet pollution air worksheeto via. 6 Oxidation Numbers Worksheet Answers | FabTemplatez www ... PDF Oxidation Number Exercise - Multidict Exercises - Give the oxidation number for the following atoms: Co(ClO) 2, CoCrO, 4 , Mg(OH) 2, RbO , 2, KMnO4= , OF, 2, KO, 2, IO! 2, Co = +2Cl = +1 Na2O2Na = +1 O = !1 , Cr = +6Co = +2 , AgNO, 3 Ag = +1 N = +5, Mg = +2 , = -2 , H2SO4,
Charting Oxidation Numbers worksheet - liveworksheets.com More Chemistry interactive worksheets. Periodic table. by yaneeam. Finding number of atoms in a chemical formula. by sriram. Covalent bond. by arnida_79. Types of Bonds (Ionic Bonding) by palmergm. Assigning Oxidation Numbers Practice - Study.com The oxidation state of {eq}N {/eq} in {eq}NH_3 {/eq} is: 20. One of the most well-known oxides of sulfur is sulfur trioxide {eq} (SO_3) {/eq}. What is the oxidation state of sulfur in this... PDF Naming Compounds - Mrs. McCutchen's Class Use the Polyatomic Ion Chart on the front of the worksheet to name these Polyatomic Ions: HCO 3 1-SO 4 2-O2 2- SO 3 2- NO 3 1-NH 4 + CrO 4 2-OH 1-PO 4 3-CO 3 2- ... Magnesium's oxidation number Sulfur's oxidation number is −1, so it will gain 1 electron. K1+ K1+ K1+ K1+ K1+ K1+ K1+ O2 ... Fillable Ionic Bonds Worksheet Answers - FormZoid Ionic Bonds Worksheet Answers can be found in a variety of formats. The most common format is an electronic worksheet that can be completed on a computer and then printed out. ... charting oxidation number worksheet, charting oxidation number chapter 7 answers, ionic bonding worksheet answers: 1 2. Related forms: Iou Template, Interim Change ...
Name That Macromolecule Worksheet Food Building Macromolecules Activity Worksheet Answers Printable Worksheets Are A Precious Classroom Tool In 2021 Macromolecules Macromolecules Activity Lab Activities . Pin On Chem . Independent Variable And Dependent Variable Worksheet Scientific Method Scientific Method Worksheet Biology Lesson Plans . Pin On Tpt Science Lessons PDF Oxidation Numbers Worksheet - brookville.k12.oh.us The oxidation number of fluorine in a compound is always -1. , Oxygen has an oxidation number of -2 unless it is combined with F (when it is +2), or it is in a peroxide (such as H2O2 or Na2O2), when it is -1. , The oxidation state of hydrogen in most of its compounds is +1 unless it is combined with a metal, in which case it is -1. , PDF bondingbasics2008 - Science Spot I , K1+ + I1- KI , Cl Mg Cl , Mg2+ + Cl1- + Cl1- MgCl2 , Mg, B6: Aluminum + Chlorine , you , see? Write the chemical , Answers will vary. , Mg2+ + I1- + I1- MgI2 , Na O Na, Na1+ + Na1+ + O2- Na2O , Cl Al Cl Cl , Al3+ + Cl1- + Cl1-+ Cl1- AlCl3 , Get Charting Oxidation Number Worksheet Answers - US Legal Forms Follow our easy steps to have your Charting Oxidation Number Worksheet Answers prepared quickly: Pick the web sample in the catalogue. Enter all required information in the required fillable fields. The easy-to-use drag&drop user interface allows you to add or relocate fields.
PDF Name: Period: CHEMISTRY: UNIT 10: Oxidation/Reduction - Weebly Assignment #1: Oxidation #s ☐ Read through slides 4-9 (online) & FILL IN blanks/charts/graphic organizers on notes sheet pg. 3-4 ☐ Watch/Use lesson videos #1-2 on website for additional notes/support (pg. 4) ☐ Complete VOCABULARY words #1-4 on vocab sheet ☐ Complete Assigning Oxidation Numbers Worksheet ! Check answer KEY - ONLINE
PDF Example Exercise 17.1 Calculating Oxidation Numbers for Carbon Calculating Oxidation Numbers for Sulfur. We can begin by recalling that the charge on an ion corresponds to the sum of the oxidation numbers. (a) In S. 2-, the oxidation number of sulfur is - 2. (b) In SO. 3 2-, the polyatomic anion has a charge of 2 -. We assign oxygen an oxidation number of - 2 and write the equation (c) In SO. 4 2-
worksheet oxidation numbers 20 Oxidation Number Worksheet With Answers | Worksheet From Home sportsbizusa.com. oxidation. Printable Periodic Tables - Science Notes And Projects sciencenotes.org. periodic table states printable oxidation chart charges labeled element pdf elements matter tables colored each bold names symbol. Oxidation Number Worksheet With Answers ...
PDF Mayfield City Schools Created Date: 10/27/2015 9:18:14 AM
PDF Oxidation and Reduction Workbook revised 1A - Laney College Use the Activity Series Chart. a) Al b) Ba2+ c) Br 2 d) Ca e) Ga3+ f) H 2 g) H+ ... Worksheet #2 Finding oxidation numbers 1. Determine the Oxidation Number of each of the elements that is underlined. a) NH 3 ... If the answer is no, write a balanced equation for the reaction that would occur. 4.
PDF Oxidation Number Exercise - Ms. Eng's Chemistry Hydrogen has an oxidation number of +1 when combined with elements onthe right side of the periodic chart (non-metals) and a -1 when combined withelements on the left side of the periodic chart (metals). Exercises - Give the oxidation number for the following atoms: HCl Cl = , HF F = (why) , NaOH O = , HI I = UH U = ,
Chapter 7.2 : Oxidation Numbers - SlideShare Chapter 7.2 : Oxidation Numbers 1. Oxidation numbersChapter 7.2 2. Objectives:List the rules for assigning oxidation numbersGive the oxidation number for each element in the formula of a chemical compound.Name binary molecular compounds using oxidation numbers and the Stock system. 3.
Get and Sign Oxidation Numbers Worksheet 2006-2022 Form Follow the step-by-step instructions below to design your oxidation numbers worksheet EMP formal form: Select the document you want to sign and click Upload. Choose My Signature. Decide on what kind of signature to create. There are three variants; a typed, drawn or uploaded signature. Create your signature and click Ok. Press Done.
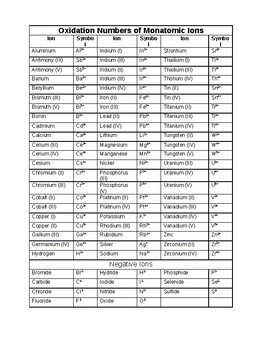
PDF VALENCE/OXIDATION NUMBERS - Texas State University Valence/Oxidation Number Potassium K 1+ Silicon Si 4+ Silver Ag 1+ Sodium Na 1+ Strontium Sr 2+ Sulfur S 2-, 4+, 6+ Tin Sn 2+, 4+ Zinc Zn 2+ Gold Au 1+, 3+ Acetate C2H3O2 1- Bromate BrO3 1- Bromic Acid HBrO3 1- Bromous Acid HBrO2 1- Bromite BrO 1- Carbonate CO3 2- Chlorate ClO3 1- Chlorite ClO2 1- Chromate CrO4 2-
oxidation numbers worksheet Balancing Equations Practice Answer Key + My PDF Collection 2021. 9 Images about Balancing Equations Practice Answer Key + My PDF Collection 2021 : Oxidation Numbers Practice Worksheet Answers - kidsworksheetfun, 34 Identifying And Balancing Chemical Equations Worksheet Answers and also Assigning Oxidation States (Example) - YouTube.
Oxidation Numbers Quiz - Softschools.com The oxidation number of oxygen in a compound is -2, except in peroxides when it is -1. 4. The oxidation number of an atom in elemental form is 0. 5. For any neutral compound, the sum of the oxidation numbers must equal 0. 6. For a polyatomic ion, the sum of the oxidation numbers must equal the ionic charge of the ion.
PDF Oxidation Numbers: Rules - Texas A&M University Oxidation Numbers: Rules 1) The oxidation number of the atoms in any free, uncombined element, is zero 2) The sum of the oxidation numbers of all atoms in a compound is zero 3) The sum of the oxidation numbers of all atoms in an ion is equal to the charge of the ion 4) The oxidation number of fluorine in all its compounds is -1
PDF RULES FOR ASSIGNING OXIDATION NUMBERS - California State University ... The oxygen appears to have "lost" 2 electrons, so its oxidation number is +2.* *When compared to the electrically neutral atom. 4. (a) The metallic element in an ionic compound has a positive oxidation number. For monoatomic cations, the oxidation number is equal to the charge on the ion. For example, Na+, Ca2+, Al 3+, Fe , etc.
Assigning Oxidation Numbers - Key - WORKSHEET - StuDocu Oxidation answers! worksheet assigning oxidation numbers name period oxidation number rules: pure element has an oxidation number of the oxidation number of an ... STI Chart SP2019; Books. Chemistry: The Central Science; ... Oxidation answers! University Syracuse University; Course Earth Science (EAR 105 ) Academic year 2016/2017;



















0 Response to "44 charting oxidation number worksheet answers"
Post a Comment